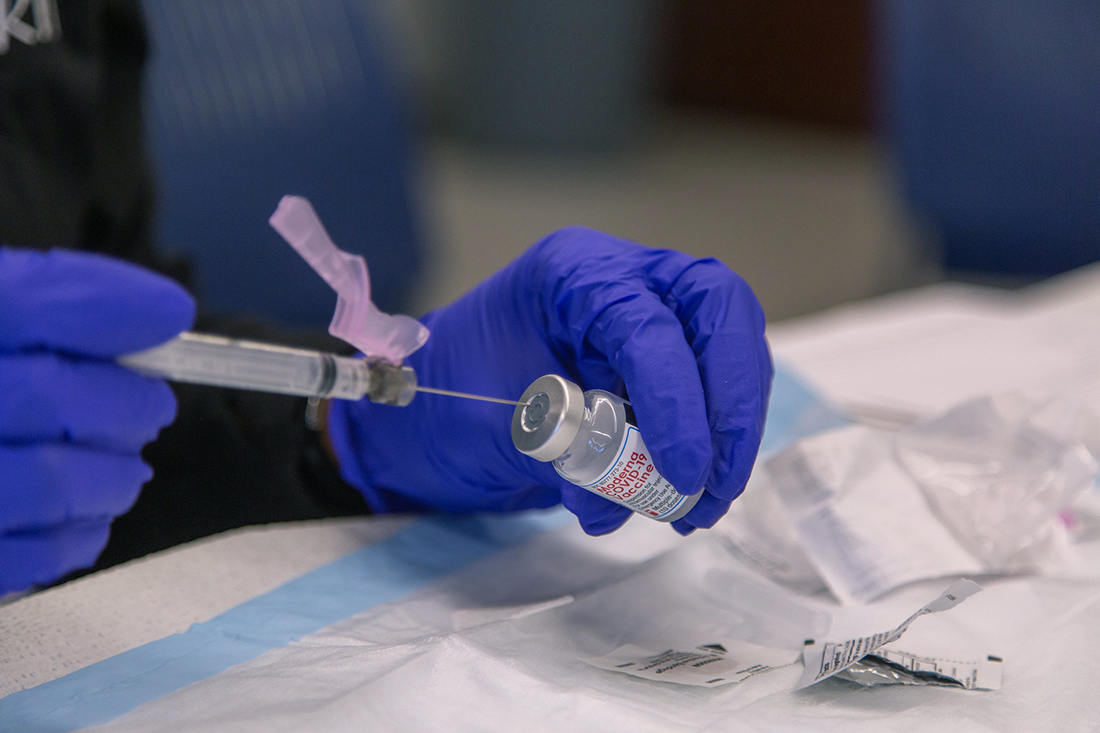
NEW HANOVER COUNTY — The simple answer to the question is: It’s not clear yet. Depending on which category you fall in, it could be in a matter of days, weeks, or even months down the line for the general public to gain access to a Covid-19 vaccine.
For now, there’s no way for the general public to set an appointment to get vaccinated. That time will come, but it’s not here yet.
Related: Facing an eviction crisis, school social workers urge families to know McKinney-Vento rights [Free]
Last week, New Hanover Regional Medical Center received its first delivery of Covid-19 vaccine, when 2,925 doses arrived at its main campus on 17th Street.
For now, only frontline health workers, first responders, nursing home staff, and seniors with two or more chronic health conditions are eligible to receive the vaccine under phase one.
There are four phases in total, with the general public falling under the last phase.
As of Tuesday afternoon, at least 2,000 staff members at NHRMC had received the vaccine, according to a hospital spokesperson. The hospital has to strategically balance how many staffers within each department are vaccinated daily, taking careful precaution not to vaccinate an entire department at once in case of unfavorable reactions.
About half of participants in vaccine trials reported modest side effects within the first few days of receiving a dose, including mile fever, headache, or fatigue — as is typical with most flu shots. However, adverse reactions are by and large not occurring in the greater population, except for in a few rare instances.
“It’s going to be a potential nine-month process before we get through the end of the final phase,” Carla Turner, assistant health director of New Hanover County said in a joint county and hospital roundtable Monday.
There’s no definitive timeline for each phase, Turner explained. The state has left the timeline up to specific localities to get through each phase before moving onto the next.
“New Hanover County might move into two before Pender County moves into phase two,” she said. “It just depends.”
Populations within the four phases, with the first phase getting the vaccine first, are being categorized as follows:
- Phase one: frontline healthcare workers; first responders; nursing home staff; seniors with two or more chronic conditions.
- Phase two: marginalized communities (including low-income individuals); seniors with one or no chronic health condition; nursing home, prison, and homeless shelter residents; non-frontline healthcare workers; critical public sector employees (education, child care, transportation, law enforcement, and fire).
- Phase three: frontline retail, electric, waste, grocery, etc. workers; public employees not already included; college students; children K-12 (if vaccines are approved for children younger than 16).
- Phase four: the general public.
All depends on supply
NHRMC Pharmacy director Dr. Mike Melroy said the hospital wants more vaccine than it’s been given at this point.
“We’re a little bit hamstrung by the supply chain for this,” he said at the virtual roundtable.
With states and municipalities clamoring for more vaccine, much of who gets what when will depend on supply. Plenty of the holdup could be administrative — storage requirements for each brand differ, while each creates its own management difficulties.
The Pfizer vaccine, the first Covid-19 option approved for emergency use by the FDA on Dec. 11, was found to be 95% effective in clinical trials. It requires an Antartic, minus-94 degrees Fahrenheit storage temperature, which limits what organizations are capable of receiving it. NHRMC is capable of storing the Pfizer vaccine and is carefully administering it now.
The FDA approved the Moderna vaccine shortly after, on Dec. 18. With a 94% effectiveness rating, Moderna can be stored in a standard refrigerator for up to 30 days, greatly expanding the organizations that can feasibly administer the vaccine.
Both options must be given in two doses. Pfzier’s second dose must be given on day 21; Moderna’s must be given on day 28.
Storage requirements and dosing differences will create administrative hurdles for healthcare facilities, but perhaps the biggest hangup, Melroy said, will ultimately come from the supply chain.
“We can set up the most efficient way to distribute this vaccine but if it’s slowly coming out of Pfizer, Moderna, and they have any kind of manufacturing hiccups or things like that, that’s going to cause delays,” he said. “A lot of what we’re able to administer is going to be what that supply looks like from these two manufacturers of these two very highly sought after vaccines.”
Distrust in the minority community
Already disproportionately impacted by Covid-19 due to pre-existing health disparities, the community that may need the vaccine the most has a legitimate reason to distrust the medical system.
Between nonconsensual sterilization and heinous research trials, minority communities have been historically subject to deceitful medical mistreatment.
Take, for instance, the Tuskeegee syphilis study. In the 1930s, more than 600 Black men were recruited to participate in a reaserch study under the promise of free medical care. Even as treatment for the disease became avaiable in the late 1940s, doctors did not give the men treatment, and instead tracked the full progression of the disease, leaving many of the men to die and suffer serious health consequences.
The legacy of events like Tuskegee has sown distrust surrounding medical trials among the minority community that last to this day.
Deborah Dicks Maxwell, president of the New Hanover County NAACP, stressed that this time around, the medical trials surrounding the Covid-19 are free from discrimination.
“There’s no discrimination of color in the trials and there’s no discrimination of color in the people receiving it,” she said. “So we need to go forward and follow the protocols for whatever New Hanover is going to dictate.”
Dr. Toren Davis, director of NHRMC Coastal Family Medical Medicine, pointed to the data from the populations that participated in the Covid-19 vaccine trails, explaining both had a fair spread of respresentation.
The Black community has experienced a 3.7 times higher hospitalization rate and 2.8 times higher death rate from Covid-19 compared to whites; Hispanics have experienced a 4.1 times higher hospitalization rate and 2.8 times higher death rate, according to Davis.
“They’re fighting the fight of health disparities as well. So how do we make sure we’re getting this out to the people that actually need it?” Davis asked. “How do we meet our patients where they are?”
Rebecca Carpenter with the Advanced Center for Covid-19 Disparities at N.C. Central University said language barrier, timely communication, and education are the top three issues for the Hispanic community.
Oftentimes vital messaging is put out late in Spanish, which leads to a delay among the Hispanic community in learning about important Covid-19 health information.
“By the time the right information gets to the Hispanic community, a lot of them miss information,” she said. “When the community doesn’t have the right information, they’re powerless.”
Getting the shot
Healthcare workers are worried that the distrust in minority communities could lead to a hesitancy in getting vaccinated.
“Is the requirement to register going to cause fear in keeping undocumented people from getting the vaccine?” Davis asked, citing registration as a big barrier to certain populations receiving care.
Vaccination requires registration. Registration involves getting participants to answer a series of questions designed to categorize them among the four phases to determine whether they’re eligible to receive the shot. “The state has a system that’s not necessarily up and running very well right now, but they’re working on it,” Turner said.
How long will the vaccine last? “We don’t know right now,” Melroy said.
According to Turner, vaccine study participants will be followed for at least two years to track effectiveness. Whether the vaccine lasts for a few months, one year, or a lifetime, is still unknown. “This stuff got to market quickly,” Melroy said. “At least I’m not 100% clear right now if this is going to be an annual type of vaccination similar to the flu shot or if this is a one and done.”
From start to finish, the vaccination process takes about thirty minutes. Participants will be monitored for at least 15 minutes after receiving the shot so that healthcare workers can observe for adverse reactions. Most negative reactions occur within this timeframe of receiving the shot.
Turner said healthcare workers should have epinephrine or Benadryl on hand in case of an allergic reaction.
“When you get the vaccine, there’s no live virus in it,” Turner explained. “What’s in the vaccine mimics the virus and makes your body think that you have the virus and it produces the antibodies and ups your immunity to that virus.”
A handful of allergic reactions have been reported since the rollout of the vaccine, but these small events shouldn’t deter the public from seeking it out when it’s available, according to Melroy.
“We’re going to see them,” he said of adverse reactions. “That’s why everybody needs to make their own decision around this. With the safety and efficacy data that has come out with the studies that really got these two vaccines to market, the benefit greatly outweighs the risk.”
Send tips and comments to [email protected]